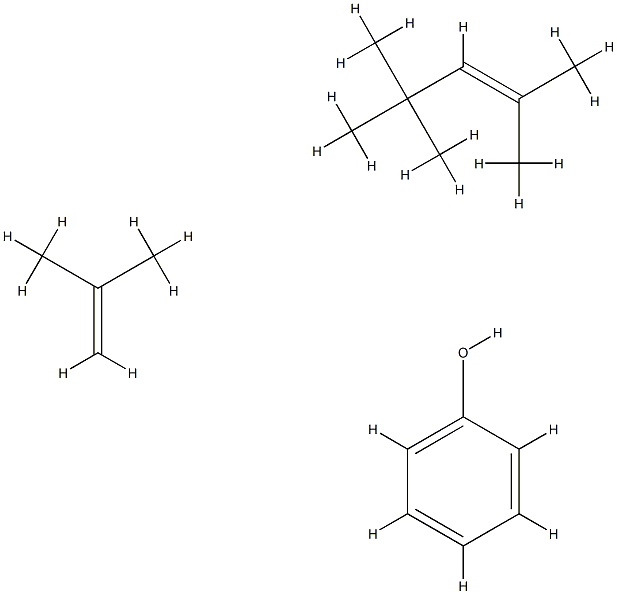
Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene
Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene
CAS: 68457-76-1
Molecular Formula: C18H30O
Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene - Names and Identifiers
| Name | Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene |
| Synonyms | Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene |
| CAS | 68457-76-1 |
| EINECS | 270-607-5 |
Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene - Physico-chemical Properties
| Molecular Formula | C18H30O |
| Molar Mass | 262.4302 |
Phenol, reaction products with isobutylene and 2,2,4-trimethylpentene - Introduction
Phenol, the reaction product of isobutene and 2,2, 4-trimethylpentene is a compound. The following is a description of the properties, uses, preparation and safety information of the compound.
Nature:
This compound is a liquid with a pungent odor. Its chemical formula is C12H18O and its molecular weight is 178.27g/mol. This compound is colorless at room temperature and has a low boiling point and melting point. It is insoluble in water, but can be dissolved in organic solvents. It is also a flammable substance.
Use:
This compound has a variety of uses in industry. It can be used as a raw material for synthetic resins and plastics, such as phenolic resins and polyesters. In addition, it can also be used in the production of fungicides, dyes and the synthesis of drugs. In the petroleum industry, it can also be used as an extractant and stabilizer.
Preparation Method:
Phenol, the reaction product of isobutylene and 2,2, 4-trimethylpentene can be prepared by hydrogenation treatment of hydrocarbons. First, isobutene and 2,2, 4-trimethylpentene are reacted together with hydrogen by heating under the action of a catalyst. A portion of the reaction product will be Phenol.
Safety Information:
This compound is irritating and corrosive. Personal protective equipment such as protective gloves, glasses and clean clothes should be worn during operation. Avoid direct contact with skin and eyes, and avoid inhaling its vapors. If accidentally contact, should immediately rinse with plenty of water, and timely medical treatment.
Nature:
This compound is a liquid with a pungent odor. Its chemical formula is C12H18O and its molecular weight is 178.27g/mol. This compound is colorless at room temperature and has a low boiling point and melting point. It is insoluble in water, but can be dissolved in organic solvents. It is also a flammable substance.
Use:
This compound has a variety of uses in industry. It can be used as a raw material for synthetic resins and plastics, such as phenolic resins and polyesters. In addition, it can also be used in the production of fungicides, dyes and the synthesis of drugs. In the petroleum industry, it can also be used as an extractant and stabilizer.
Preparation Method:
Phenol, the reaction product of isobutylene and 2,2, 4-trimethylpentene can be prepared by hydrogenation treatment of hydrocarbons. First, isobutene and 2,2, 4-trimethylpentene are reacted together with hydrogen by heating under the action of a catalyst. A portion of the reaction product will be Phenol.
Safety Information:
This compound is irritating and corrosive. Personal protective equipment such as protective gloves, glasses and clean clothes should be worn during operation. Avoid direct contact with skin and eyes, and avoid inhaling its vapors. If accidentally contact, should immediately rinse with plenty of water, and timely medical treatment.
Last Update:2024-04-09 21:01:54